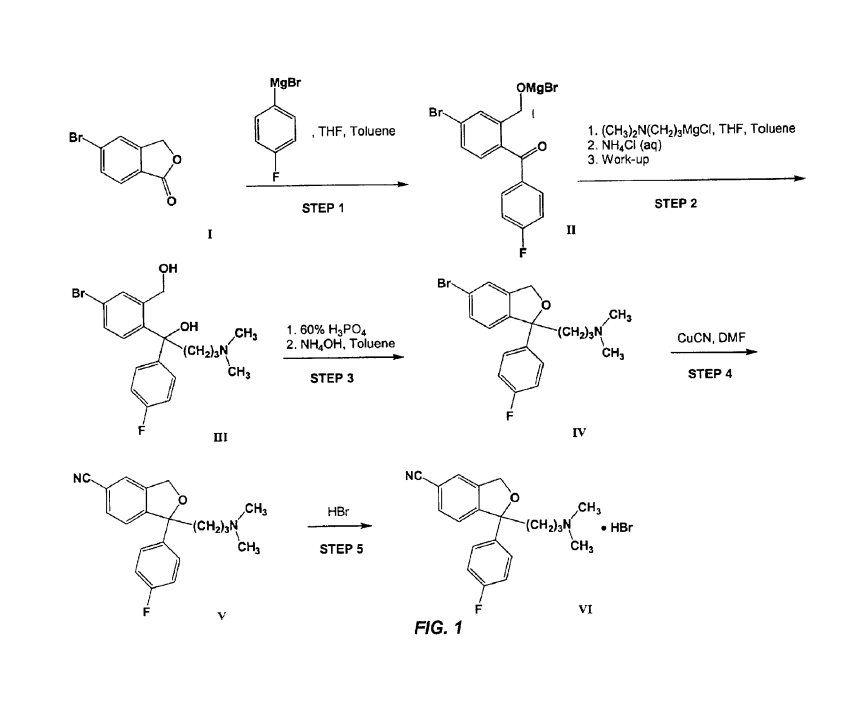
Synthesis of Citalopram
The synthesis of commerical Citalopram is a very complicated process in actuality. In fact, pharmaceutical quality Citalopram requires the material used to have a purity in excess of 99.7%. However, on paper it can be 'dumbed-down' to a 5-step conversion of 5-bromophthalide to Citalopram. The following procedures are the synthesization process described in the original patent for Citalopram. A picture will be provided for reference. All the information given is referenced by [5].
Step 1: A compound of Formula I ("5-BP") is reacted with p-fluorophenyl-magnesium halide.
Step 2: The intermediate of a compound of Formula II is isolated and reacted with N,Ndimethylaminopropylmagnesium halide to give the diol of Formula III ("Br-Diol"). The Grignard reaction is conducted in traditional solvents, such as diethyl ether and THE.
The work-up involves the following steps:
The reaction mixture is quenched into ice water;
An aqueous saturated ammonium chloride solution is added;
The mixture is extracted with diethyl ether;
The ether phase is then extracted with 20% aqueous acetic acid;
The acid phase is made alkaline with 10 N aqueous sodium hydroxide;
The aqueous phase is extracted with diethyl ether (2x);
The combined ethereal extracts are dried over anhydrous K2CO3;
The ether extract is treated with activated carbon;
And the solvent is evaporated in a vacuum to give Br-Diol, an oil.
It is noted that Br-Diol is isolated as the free amine and is an oil. The physical characteristics of Br-Diol are important. Since Br-Diol is an oil, it cannot be isolated as a crystalline solid and, thus, it cannot be purified by techniques such as crystallization/ re-crystallization. Purification of this oil by crystallization or similar techniques is never mentioned in documents detailing it's synthesis. In addition, in order to meet the tight specifications for Citalopram, it is critical that purity is established at this stage.
Step 3: Br-Diol is subjected to a ring closure reaction with 60% aqueous phosphoric acid. In a typical reaction, 5-bromophthalide is heated with excess (30 equivalents) 60% aqueous phosphoric acid for 3 hhours and then neutralized with saturated aqueous ammonia. The resulting mixture is then extracted with diethyl ether, and the ether extract is dried over potassium carbonate. The ether extract is then treated with activated charcoal and stripped of solvent under reduced pressure to give the compound of Formula IV ("5-Br").
Step 4: 5-Br is reacted with cuprous cyanide in DMF to give, after the work-up, Citalopram. The reaction conditions and work-up for the process are described as follows:
5-Br is reacted with CuCN in DMF at reflux for 4 hours;
The reaction mixture is cooled to 55° C. and quenched into an aqueous solution of ethylene diamine;
The oily layer is separated and the aqueous layer is extracted with benzene;
The combined organic phases are washed with 10% aqueous sodium cyanide;
The organic layer is dried, treated with activated carbon, and concentrated under vacuum to give an oil;
The oil is dissolved in ether and extracted with aqueous acetic acid;
The acetic acid layer is made alkaline with 10 N aqueous 5 sodium hydroxide and extracted with ether;
And the ethereal extract is dried over K2CO3 , treated with activated charcoal, and stripped of solvent to give Citalopram.
Unfortunately, there are numerous problems with step four in actuality. First, the reaction does not go to completion in 4 h. In reality, conversion after 4 h is <10%. Removal of unreacted 5-Br is difficult and normal purification techniques, such as extraction, crystallization, etc., are not effective. When the reaction is pushed to achieve higher conversion, formation of numerous unidentified side-products is observed.
Step 5: In step five Citalopram is converted to Citalopram·HBR, in order to be used for commercial use, by adding HBR.
An alternative synthesis of Citalopram has also been described, albeit in less detail:
This process is based on 5-cyanophthalide (,,5-CN").
In this process, 5-CN is reacted with 4-fiuorophenylmagnesium halide and N,Ndimethylaminopropylmagnesium halide to give the corresponding hydroxy intermediate that is then dehydrated with 30 sulfuric acid to give Citalopram.
Step 2: The intermediate of a compound of Formula II is isolated and reacted with N,Ndimethylaminopropylmagnesium halide to give the diol of Formula III ("Br-Diol"). The Grignard reaction is conducted in traditional solvents, such as diethyl ether and THE.
The work-up involves the following steps:
The reaction mixture is quenched into ice water;
An aqueous saturated ammonium chloride solution is added;
The mixture is extracted with diethyl ether;
The ether phase is then extracted with 20% aqueous acetic acid;
The acid phase is made alkaline with 10 N aqueous sodium hydroxide;
The aqueous phase is extracted with diethyl ether (2x);
The combined ethereal extracts are dried over anhydrous K2CO3;
The ether extract is treated with activated carbon;
And the solvent is evaporated in a vacuum to give Br-Diol, an oil.
It is noted that Br-Diol is isolated as the free amine and is an oil. The physical characteristics of Br-Diol are important. Since Br-Diol is an oil, it cannot be isolated as a crystalline solid and, thus, it cannot be purified by techniques such as crystallization/ re-crystallization. Purification of this oil by crystallization or similar techniques is never mentioned in documents detailing it's synthesis. In addition, in order to meet the tight specifications for Citalopram, it is critical that purity is established at this stage.
Step 3: Br-Diol is subjected to a ring closure reaction with 60% aqueous phosphoric acid. In a typical reaction, 5-bromophthalide is heated with excess (30 equivalents) 60% aqueous phosphoric acid for 3 hhours and then neutralized with saturated aqueous ammonia. The resulting mixture is then extracted with diethyl ether, and the ether extract is dried over potassium carbonate. The ether extract is then treated with activated charcoal and stripped of solvent under reduced pressure to give the compound of Formula IV ("5-Br").
Step 4: 5-Br is reacted with cuprous cyanide in DMF to give, after the work-up, Citalopram. The reaction conditions and work-up for the process are described as follows:
5-Br is reacted with CuCN in DMF at reflux for 4 hours;
The reaction mixture is cooled to 55° C. and quenched into an aqueous solution of ethylene diamine;
The oily layer is separated and the aqueous layer is extracted with benzene;
The combined organic phases are washed with 10% aqueous sodium cyanide;
The organic layer is dried, treated with activated carbon, and concentrated under vacuum to give an oil;
The oil is dissolved in ether and extracted with aqueous acetic acid;
The acetic acid layer is made alkaline with 10 N aqueous 5 sodium hydroxide and extracted with ether;
And the ethereal extract is dried over K2CO3 , treated with activated charcoal, and stripped of solvent to give Citalopram.
Unfortunately, there are numerous problems with step four in actuality. First, the reaction does not go to completion in 4 h. In reality, conversion after 4 h is <10%. Removal of unreacted 5-Br is difficult and normal purification techniques, such as extraction, crystallization, etc., are not effective. When the reaction is pushed to achieve higher conversion, formation of numerous unidentified side-products is observed.
Step 5: In step five Citalopram is converted to Citalopram·HBR, in order to be used for commercial use, by adding HBR.
An alternative synthesis of Citalopram has also been described, albeit in less detail:
This process is based on 5-cyanophthalide (,,5-CN").
In this process, 5-CN is reacted with 4-fiuorophenylmagnesium halide and N,Ndimethylaminopropylmagnesium halide to give the corresponding hydroxy intermediate that is then dehydrated with 30 sulfuric acid to give Citalopram.